Health
MIT Scientists Uncover Surprising Genomic Loops During Cell Division
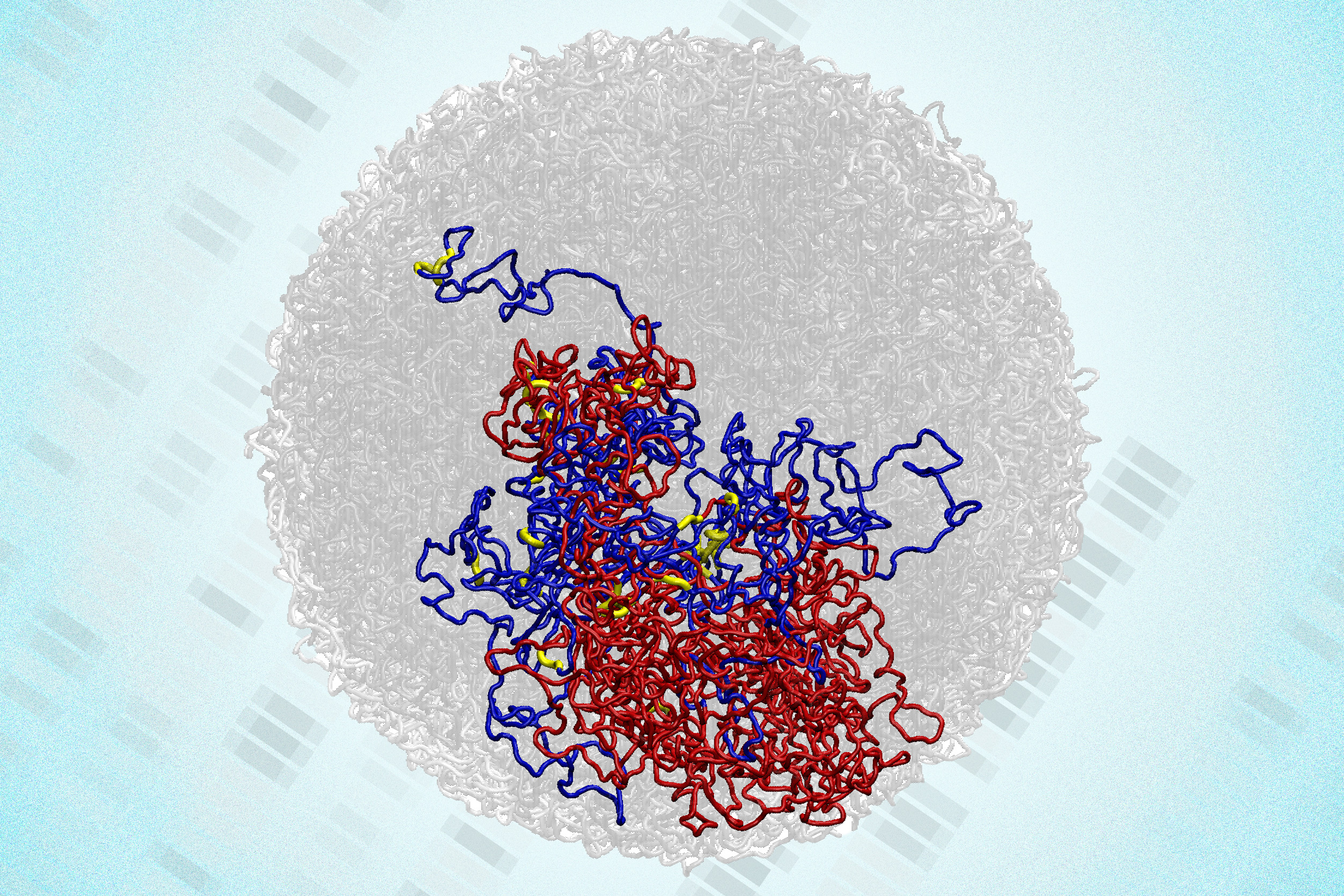
Researchers at the Massachusetts Institute of Technology (MIT) have made a groundbreaking discovery about the structure of genomes during cell division. Their study, published in Nature Structural and Molecular Biology, reveals that tiny 3D loops within the genomes of dividing cells persist, contradicting long-held beliefs about the genome’s behavior during mitosis.
Traditionally, scientists thought that as cells divide, they lose their intricate 3D structure, which plays a crucial role in gene regulation. This new research indicates that instead of vanishing, small loops that connect regulatory elements and genes remain intact during this process. Anders Sejr Hansen, an associate professor of biological engineering at MIT, explains, “This study really helps to clarify how we should think about mitosis. In the past, mitosis was thought of as a blank slate, with no transcription and no structure related to gene activity. Now we know that’s not quite the case; there’s always structure. It never goes away.”
The research team utilized a high-resolution genome mapping technique called Region-Capture Micro-C (RC-MC), which allows for examination of 3D genome structures with unprecedented detail. This method, developed in 2023, provides a resolution 100 to 1,000 times greater than previous techniques. During their investigation, the team discovered new structures they named “microcompartments.” These tiny, highly connected loops form when nearby enhancers and promoters adhere to each other, facilitating interactions that can influence gene expression.
Hansen, along with lead author Viraat Goel, noted the significance of these findings in understanding how cells maintain regulatory structures during division. “The findings help to bridge the structure of the genome to its function in managing how genes are turned on and off, which has been an outstanding challenge in the field for decades,” Goel stated.
The research also uncovered that these regulatory loops tend to strengthen as chromosomes compact in preparation for division. This compaction facilitates closer proximity between genetic regulatory elements, which may help cells “remember” interactions from one cycle to the next. Edward Banigan, a research scientist involved in the study, contributed to the exploration of this novel genomic landscape.
Over the last two decades, scientists have recognized that DNA organizes itself into 3D loops within the cell nucleus, enabling interactions between distant genes and regulatory regions. Prior mapping techniques, primarily using Hi-C, lacked the resolution to identify all specific interactions. The newly developed RC-MC technique addresses these limitations, leading to the discovery of microcompartments that differ from previously identified genome structures.
The research team initially believed that microcompartments would disappear during mitosis, as larger genome structures like A/B compartments and topologically associating domains (TADs) do. Contrary to their expectations, they found that microcompartments not only persisted but became more pronounced during cell division. This unexpected finding challenges established notions of gene regulation during mitosis.
“This study leverages the unprecedented genomic resolution of the RC-MC assay to reveal new and surprising aspects of mitotic chromatin organization,” commented Effie Apostolou, an associate professor of molecular biology in medicine at Weill Cornell Medicine, who was not involved in the research.
Another intriguing aspect of the study indicates that these microcompartments may play a role in the transcriptional spike observed at the end of mitosis. Historically, it was believed that transcription halted entirely during this phase. However, recent studies have shown brief spikes in transcription activity, which the MIT researchers linked to the presence of microcompartments near genes that exhibit this behavior.
Hansen highlighted that the environment created during mitosis could inadvertently promote the formation of these loops, which the cell may then prune away once it enters the G1 phase. “It almost seems like this transcriptional spiking in mitosis is an undesirable accident that arises from generating a uniquely favorable environment for microcompartments to form during mitosis,” he explained.
The implications of this research extend beyond mere academic interest. As chromosomes can also be influenced by a cell’s shape and size, the team plans to investigate how these variations affect genomic structure and gene regulation. “We are thinking about some natural biological settings where cells change shape and size, and whether we can perhaps explain some 3D genome changes that previously lacked an explanation,” Hansen added.
This pioneering study received funding from several reputable sources, including the National Institutes of Health and the National Science Foundation. The findings pave the way for future research into the complexities of gene regulation and chromosomal organization during cell division.
-

 Science3 months ago
Science3 months agoUniversity of Hawaiʻi Joins $25.6M AI Project to Enhance Disaster Monitoring
-

 Top Stories3 months ago
Top Stories3 months agoJoleen Chaney, Beloved Journalist, Passes Leaving Lasting Legacy
-

 Top Stories3 months ago
Top Stories3 months agoUrgent Update: Tom Aspinall’s Vision Deteriorates After UFC 321
-

 Health2 months ago
Health2 months agoMajor Grant Enhances Cancer Care and Research in Hawaiʻi
-

 Business3 months ago
Business3 months agoTrulioo Introduces KYA to Revolutionize Digital Verification
-

 Top Stories4 months ago
Top Stories4 months agoAI Disruption: AWS Faces Threat as Startups Shift Cloud Focus
-

 Entertainment4 months ago
Entertainment4 months agoDiscover the Full Map of Pokémon Legends: Z-A’s Lumiose City
-

 Science4 months ago
Science4 months agoTime Crystals Revolutionize Quantum Computing Potential
-

 World4 months ago
World4 months agoHoneywell Forecasts Record Business Jet Deliveries Over Next Decade
-

 Entertainment4 months ago
Entertainment4 months agoParenthood Set to Depart Hulu: What Fans Need to Know
-

 Sports4 months ago
Sports4 months agoMel Kiper Jr. Reveals Top 25 Prospects for the 2026 NFL Draft
-

 Top Stories4 months ago
Top Stories4 months agoGOP Faces Backlash as Protests Surge Against Trump Policies